 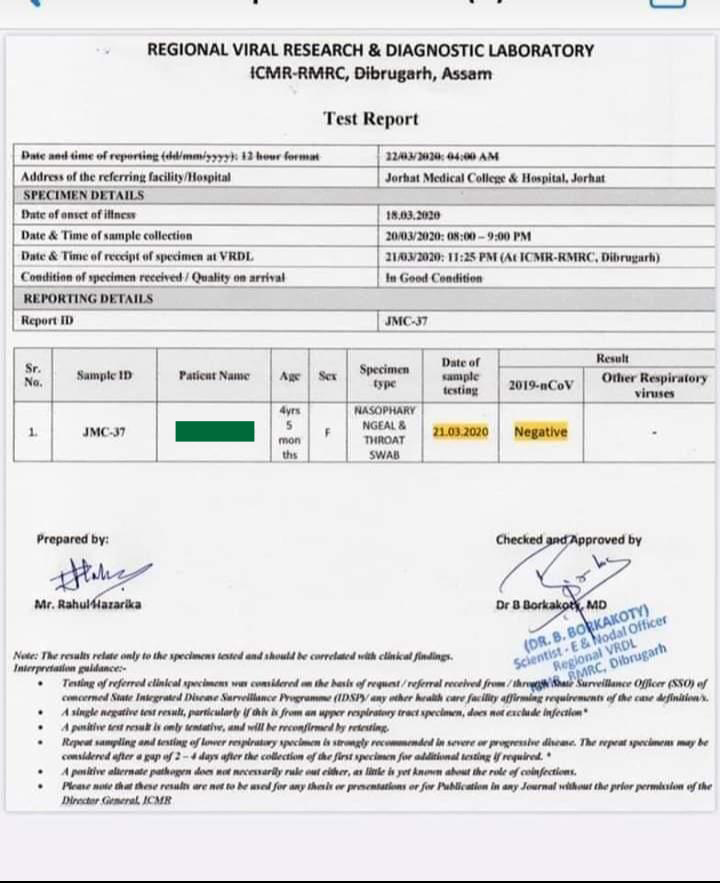
The table below shows the required reportable data components that each entity must report under the Board’s emergency rule. Emails will be sent out as needed and likely be between one to two emails per month. Monthly listening sessions are presented, and recorded, by the ELR team. Contact you're interested in this time-saving measure.Īdditionally, the ELR team has created a listserv to provide general information related to WA-specific electronic laboratory reporting, national updates, frequently asked questions and future listening sessions. Labs cannot make that determination on their own. The Department has a process to work with and approve labs for submitting their results electronically and satisfying their disease reporting requirements. Once electronic reporting to the Department is in place, labs do not need to send additional reports to local health jurisdictions (LHJs) or the Centers for Disease Control and Prevention (CDC). The table below describes required data components for each reporting entity.Įlectronic laboratory reporting (ELR) to the Department meets local, state, and federal lab reporting requirements for COVID-19 results. Laboratories must also include certain data when submitting specimens to the Department and when referring specimens to other laboratories. LabsĮntities licensed to conduct moderate or high complexity testing must report all positive, negative, and inconclusive test results from all nucleic acid amplification tests (NAAT) and antigen tests performed for COVID-19 to their local health jurisdiction. More information on how to report can be found here. The table below describes required data components for each reporting entity. POC testing sites are required to meet the reporting requirements of laboratories for positive cases. Point-of-Care TestingĮntities licensed to conduct waived tests under a certificate of waiver, including point-of-care (POC) testing for COVID-19, must report all positive COVID-19 test results to their local health jurisdiction. The Department is working on a process for electronic case reporting and will announce when it is available. 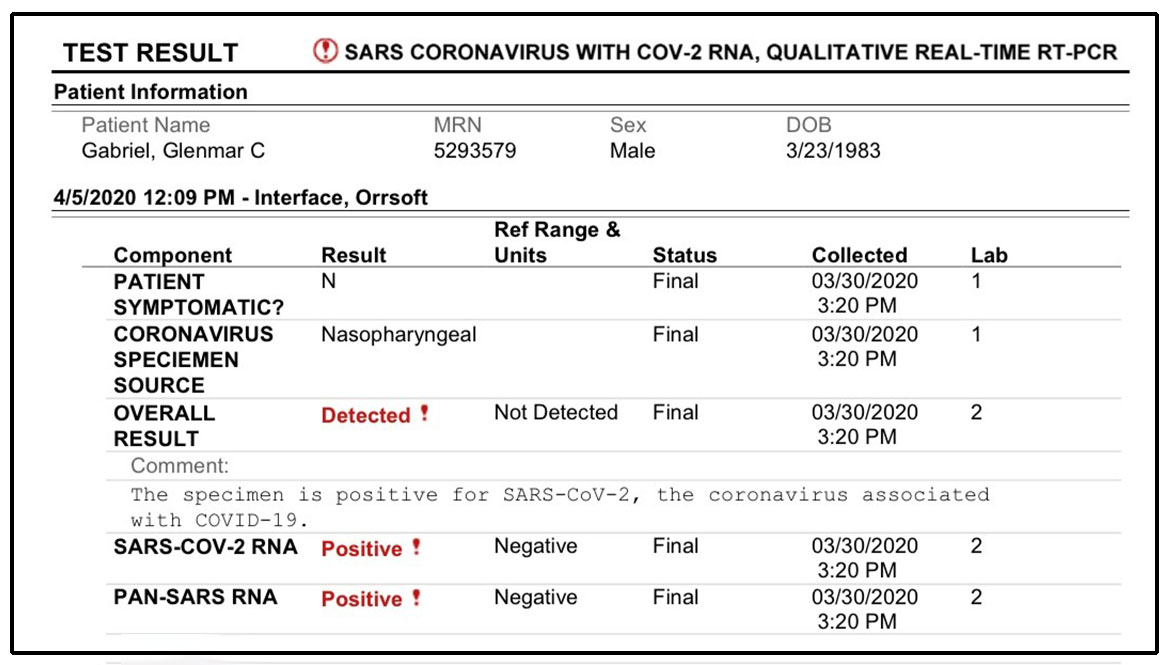
Health care providers and facilities must include certain patient demographic and ordering facility data in their laboratory orders and case reports. Health care providers, or health care facilities on behalf of providers, must submit individual case reports of COVID-19 to their local health jurisdiction. The Washington State Department of Health (Department) is working with labs, providers, and local health jurisdictions to implement provisions in the emergency rule. The Washington State Board of Health (Board) recently adopted a seventh emergency rule to ensure continued compliance of these federal requirements in alignment with chapter 246-101 WAC, Notifiable Conditions. Department of Health and Human Services (HHS) issued reporting requirements for laboratories to ensure complete demographic data reporting with COVID-19 test results. In response to the COVID-19 pandemic, the U.S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
